Properties of Sand
Important Point
It can be used as soon as it is obtained from the source. Contains binding content (5-20 per cent), water (5-8 per cent) and a significant amount of organic matter. The moisture content should be preserved for a long time.
Finishing on natural sand moulds is fine. It’s cheaper than most sands. It has less refractoriness. It is used for casting CI and non-ferrous metals. Natural sand moulds can be quickly repaired.
When combined with bentonite, the properties of sand are enhanced and have properties like Synthetic Sand.
1. Physical Properties of Sand
Here the different types of physical proprietors of sand are as follows with full detail.
- Grain Size of Sand
- Grain Shape of Sand
- Shape and Distribution of Sand Grains
- Size Distribution of Sand
a. Grain Size of Sand
The grain shape and scale of the moulding sand determine the overall surface area of the grains found in the unit mass. Total surface area is defined as a particular surface area. A particular surface provides a rough understanding of the quantity of binder required to coat grains of sand moulding.
Grain size and distribution affects many properties of sand, such as permeability, flowability, refractory properties, surface fineness and strength. The finer the grains of sand, the finer the entire sand. Fine grain sands offer a good surface finish but have poor permeability.
With the same content of clay, the environmental strength is greater in the context of fine sand than in the context of rough sands. Solid and evenly graded sand has high permeability, decent reactiveness as well as high flowability. Usually, foundry sand has grain sizes around 0.1 and 1.0 mm.
Fine-grained sands are being used to the importance of ensuring as well as small castings. Large grained sands are being used to produce large castings.
Also, read: What Is Honeycomb In Concrete | Cause | Cure | Type of Grouting
b. Grain Shape of Sand
The shape of the grain is defined in terms of angularity as well as sphericity. The grains of sand range from well rounded, subrounded, angular and also very angular. Inside each angularity unit, grains can be of a high, medium, or low sphericity. The angularity of sand is measured by visual inspection using a low-power microscope.
The best foundry sands include grains that have been rounded with medium to high sphericity, offering goof flowability and high-intensity permeability at low binder additions. More angular and lower spherical sands require higher binder additions, lower packing density and poorer flowability.
Also, read: What Is Honeycomb In Concrete | Cause | Cure | Type of Grouting
c. Shape and Distribution of Sand Grains
The texture, size and form of the sand grains are critical for controlling the consistency of the mould. Many of the mould aggregates are mixtures of fresh sand and recycled sand, which comprise not only reclaimed sand but also core sands. In deciding the scale, shape and distribution of sand grains, it is necessary to note that the grain shape corresponds to the amount of sand surface area and that the distribution of grain size influences the permeability of the mould.
If the sand surface area increases, the volume of bonding content (usually clay and water) must increase if the sand is to be adequately bonded. As a result, a difference in surface area, possibly due to a change in sand form or the proportion of core sand being retrieved, would result in a related change in the amount of bond required.
Rounded grains have a low surface-area-to-volume ratio and are thus favoured for core processing because they need the least amount of binder. However, when recycled into the sand moulding system, their form can be disadvantageous if the moulding system usually uses a high percentage of clay and water to allow quick, automatic moulding. This is because the rounded grains need less sand than the rest of the structure.
Angular sands have the highest surface area (with the exception of sands that crack quickly and contain a significant proportion of small grains and fine sands) and thus require more mulling, bonding and moisture. The angularity of sand rises with its use when the sand is broken by thermal or mechanical shocks.
The porosity of the mould regulates its permeability, i.e. the capacity of the mould to allow the gasses produced during the spilling to pass through all the mould. The maximum porosity is attributed to grains that are all about the same size.
As the size range widens, there are plenty more particles that are tiny enough just to occupy the gap between large grains. When grains break down by constant recycling, there seem to be smaller grains as well as the porosity of the mould reduces.
Furthermore, if the porosity of a mould is too high, the metal will penetrate sand grains and create a burn-in flaw. It is also important to balance the delivery of base sand and proceed to screen the sand and then use dust collectors throughout recycling to extract fines and to decide the correct bond addition.
Also, read: What Is Slump Cone Test | Principle of Slump Test | Types of Concrete Slump
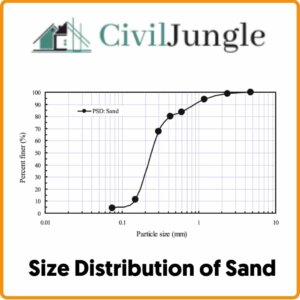
d. Size Distribution of Sand
The size distribution of sand influences the consistency of the casting. Coarse grain sand allows the metal to enter the moulds and cores, giving the castings a weak surface finish. Fine-grained soils produce a better surface finish but need a higher bonding content and low permeability can cause a gas defects in castings.
2. Chemical Properties of Sand
Here the different types of physical chemicals of sand are as follows with full detail.
- Soil Reaction (PH) of Sand
- Silicate Clay Presence in Sand
- Cation Exchange Capacity (CEC) in Sand
a. Soil Reaction (PH) of Sand
By reference, “PH” is a calculation of the concentration of active hydrogen ions (H+). It is an indicator of the acidity or alkalinity of the soil and is therefore defined as the “soil reaction.”
The pH varies from 0 to 14, for values under 7.0 acid as well as values above 7.0 alkaline. The pH value of 7 is known to be neutral, where H+ and OH-are equivalent, all at a concentration of 10-7 moles/liter. The pH of 4.0 is ten times more acidic than the pH of 5.0.
The most significant influence of ph in the soil is on ion solubility, and this, in turn, determines microbial growth including plant growth. The initial ph of 6.0 to 6.8 is suitable for many of these plants as it fits the optimal solubility of the most essential plant nutrients. Few minor elements (e.g. iron) yet most toxic chemicals are much more soluble at reduced PH. This allows pH management essential for regulating the transport of toxic metals (and possible contaminated groundwater) in soil.
With construction and also an industrial perspective, it is important to decide the pH of the soil mass. The acidic, as well as basic essence of the soil, should be understood for growing healthy crops. In the other side, in building work, strongly acidic soils would have a bearing on the road’s bitumen stability and detrimental effects on concrete strength. (The salinity of soils can also raise cost of maintenance)
Hydrogen, as well as aluminium, become dominant interchangeable cations in acid soils. The latter would be soluble within acidic conditions, and its reactivity with water (hydrolysis) creates hydrogen ions. Calcium, as well as magnesium, are simple cations; the relative sum of acidic cations increases as their concentrations increase.
Factors influencing soil pH include parent material, vegetation and atmosphere. Some rocks and sediments contain more acidic soils than others: quartz-rich sandstone is acidic; limestone is alkaline. Some types of vegetation, especially conifers, produce organic acids that can contribute to lower soil pH values. In wetlands such as the eastern US, soils begin to become more acidic with time, because runoff washes out essential cations and replaces them with hydrogen.
Also, read: What Is Cover in Concrete | Clear Cover in Beams, Slab, Column, Footing
b. Silicate Clay Presence in Sand
The presence of silicate clay content influences the chemical properties of the soil mass. Clay particles have a wide surface area and are the best materials in the soil mass. Clay particle increases the reactivity of the soil mass and influences the cohesion of the soil mass by forming compounds with foreign materials.
The assessment of the occurrence of silicate clay is necessary in order to determine the reactivity of the soil mass and its consistency with the mixtures and building materials used in concrete.
Also, read: What Is DLC (Dry Lean Concrete) | Advantge of DLC (Dry Lean Concrete )
c. Cation Exchange Capacity (CEC) in Sand
Any plant nutrients and metals occur in the soil atmosphere as positively charged ions or “cations.” Among the most important soil cations are hydrogen (H+), aluminium (Al+3), calcium (Ca+2), magnesium (Mg+2) and potassium (K+). Many heavy metals also occur in the soil ecosystem as cations.
Clay and organic matter particles are primarily negatively charged (anions) and have the capacity to keep cations from being “leached” or washed away. Adsorbed cations are subject to substitution by other cations in a quick, reversible mechanism called cation exchange.
The Cation Exchange Capacity of the soil is indeed an estimate of the maximum of the negative value at a given weight of the sample, or the sum of cations that a given soil sample can carry in an interchangeable shape.
Cations escaping the exchange sites meet the soil particles where they’ll be absorbed by plants, interact with several other soil elements, or transported with runoff water.
The higher the quantity of clay as well as organic matter much higher the Cation Exchange Capacity, while various forms of clay minerals and organic matter may differ in Cation Exchange Capacity.
Cation exchange is an important process in soils for the preservation and delivery of plant nutrients and for the adsorption of pollutants. It plays an important function in soil wastewater treatment. Sandy soils with low CEC are usually unsuitable for septic systems because they have no adsorptive capability and there is a potential for groundwater.
What Is Sand?
Sand is a granular material composed of finely divided rock and mineral particles. Sand has various compositions but is defined by its grain size. Sand can also refer to a textural class of soil or soil type; i.e., a soil containing more than 85 percent sand-sized particles by mass.
Properties of Sand
Sand is a granular material composed of finely divided rock and mineral particles. Sand has various compositions but is defined by its grain size. Sand grains are smaller than gravel and coarser than silt.
What Is Sand and Its Properties?
Sand is a granular material composed of finely divided rock and mineral particles. Sand has various compositions but is defined by its grain size. Sand grains are smaller than gravel and coarser than silt. Desert sand, although plentiful, is not suitable for concrete.
How Can You Tell the Quality of Sand?
Take a Sample of sand and add it in Sodium Hydroxide [NaOH] Solution, Stir the solution for few minutes, if the color of solution changes to brown, then the sand has organic impurities which are not suitable for construction. Good quality of sand shows lighter color when it is mixed with NaOH solution.
What Makes Good Building Sand?
Flowability, green strength and dry strength are few of the characteristic properties of moulding sand, but not hardenability.
Properties of Good Sand
- Should be completely inert.
- Grains should be sharp, strong & angular.
- Should not contain any hygroscopic salts.
- Should not contain clay & silt; usually 3-4% clay & silt is ordinarily permitted for practical reasons.
- There should be no organic matter.
Quality of Good Sand
The grains should be sharp, angular and coarse. The sand should be free from clay material and organic matters. The grains should be of durable minerals. It should be free from salts.
Chemical Properties of Sand
The main element of quartz sand is silicon dioxide (SiO2). The oxygen atoms form with the silicon atom in the centre a tetrahedral structure. Each oxygen atom simultaneously belongs to two silicon atoms. In this manner the tetrahedral structures have a cross-linked high-molecular weight.
Characteristics of Sand
Sand soils are often dry, nutrient deficient and fast-draining. They have little (or no) ability to transport water from deeper layers through capillary transport.
Sand Properties / Sand Physical Properties
- Should be completely inert.
- Grains should be sharp, strong & angular.
- Should not contain any hygroscopic salts (i.e., CaCl2, MgCl2, etc.).
- Should not contain clay & silt; usually 3-4% clay & silt is ordinarily permitted for practical reasons.
- There should be no organic matter.
Property of Sand / What Are the Properties of Sand?
Grains should be sharp, strong & angular. Should not contain any hygroscopic salts (i.e., CaCl2, MgCl2, etc.). Should not contain clay & silt; usually 3-4% clay & silt is ordinarily permitted for practical reasons. There should be no organic matter.
Characteristic of Sand
Sand soils are often dry, nutrient deficient and fast-draining. They have little (or no) ability to transport water from deeper layers through capillary transport.
Physical Properties of Sandy Soil
Sandy soils are often considered as soils with physical properties easy to define: weak structure or no structure, poor water retention properties, high permeability, highly sensitivity to compaction with many adverse consequences.
Like this post? Share it with your friends!
Suggested Read –
- Sand Chemical Composition
- Concrete Material Calculation / Concrete Quantity
- Definition Efflorescence | Efflorescence in Concrete
- What is Plum Concrete | Application | Mix Design | Methodology
- Difference Between One Way Slab and Two Way Slab | What is Slab
- How to Calculate Staircase | Concrete & Bar Bending Schedule (BBS) | Staircase Reinforcement Details
- Building Layout | How to Building Layout | What Is Method of Layout of Building | Control Lines of Construction | Construction Layout
- What Is Road Pattern | Different Types of Road Patterns | Grid Pattern Definition | Radial Pattern | Rectangular or Block Pattern | Minimum Travel Pattern
- Definition of Shear Force and Bending Moment | What Is Shear Force | What Is Bending Moment | Relation Between Loading, Shear Force & Bending Moment








Leave a Reply